U60: Metal Free Organic Sensitizer
Summary
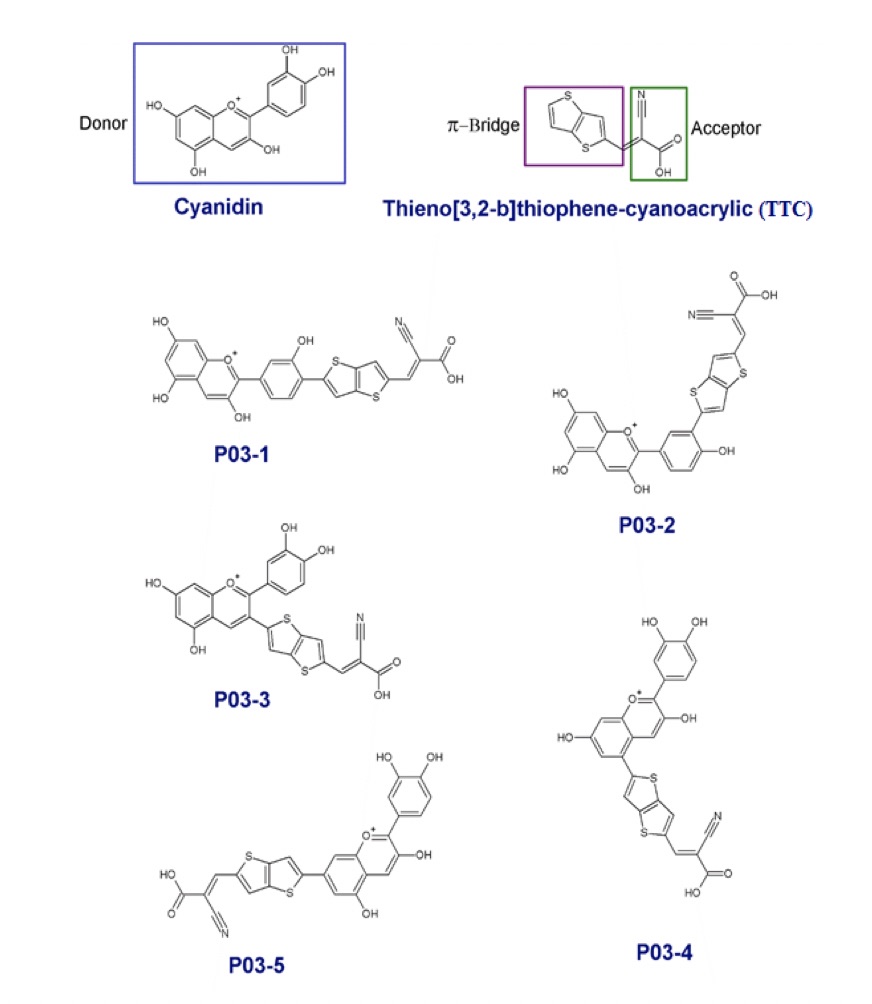
The invention relates to a metal-free organic photosensitizer and their use in solar cell making. It relates to natural photosensitizers with absorption spectra extending up to near infrared region (IR). The invention is capable of achieving a broader absorption spectrum that covers the entire visible range and extends up to the near-IR region of 2500 nm. The present disclosure provides a series of new organic D−π−A architected photosensitizer that is derived from cyanidin, which is abundantly available in nature. Cyanidin acts as the donor group (D) while cyanoacrylic acid and thieno [3, 2-b] thiophene are employed as the acceptor (A) and π-spacer, respectively. Sensitization performance calculated by density functional theory (DFT) and time-dependent density functional theory (TDDFT) concludes to a plurality of preferred substitutions of π-A combination to cyanidin molecule thereby leading to an efficient dye-sensitized solar cell (DSSC). Such π-A substitutions result in a broader absorption spectrum with a redshift up to 2500 nm.
The invention relates to a metal-free organic photosensitizer and their use in electricity generation and related equipment, more particularly, it discloses a series of organic photosensitizers designed by combination of an electron-rich natural dye as the electron donor (D) group, a promising electron acceptor as the electron acceptor (A) group, and π-spacer (π) moiety. The novel photosensitizers disclosed herein exhibit surprising broader absorption spectra extending up to near infrared region (IR).
Status
UBD Ref No: U60 Status: Filed
Inventors: Piyasiri Ekanayake, Kalpana Galappaththi, Mohammad Iskandar Petra
Documents: BN/N/2018/0029